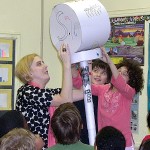Copper
October 6, 2011 in Elements
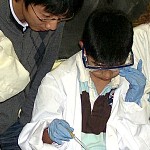
The aging effects of oxidation were revealed to Ms Reavell’s 4th grade class at École FACE School on Friday the 17th of April, 2009, thanks to MLP Team Copper (Cu), Jessie Trubiano (Bachelor student of Art Education, Concordia) and David Sabatino (PDF U Montréal, PhD McGill ’07). Showing images of the Statue of Liberty, Jessie displayed how the brown face of the copper statue of Ms liberty has turned to green over time as her copper complexion reacted with oxygen and converted to copper oxide. To put back the shine in the faces of old pennies, David placed them in vinegar and the students watched as the copper oxide was digested away from the metal surface into the acetic acid solution leaving shiny pennies. The students watched next the reduction of the clear solution containing the oxidized copper by using iron nails, onto which the brown copper plated. Through a discussion of the applications of copper in everyday life, from our plumbing, to our electrical wiring, to the coins in our pockets (albeit the present penny is only 4.5% copper), to the making of bronze (an alloy of copper and tin), the students gained a greater appreciation of the importance of this essential element.
In recognition of the Copper badges worn by North American policemen, which has been claimed for the etymology of words in phrases such as ”cops and robbers” and ”Come an’ get me, copper!” (see “Angels With Dirty Faces”) the students created their own badges by sculpting with copper wire. Although ”Cop” (policeman) probably comes from “cop,” meaning to “nab” a suspect, a myth without factual basis suggests that “cop” comes from “copper,” the copper badge that the police wore. The 4 March 1976, Wall Street Journal cites: The New York City Police Museum proudly claims that its 1845 copper badge was the origin of the slang terms “cop” and “copper,” see: http://www.barrypopik.com/index.php/new_york_city/entry/cop_copper_badge_myth/
Bending metal as our ancestors once did in the “Copper Age”, grateful for an electrochemical MLP experience, the students thanked Team Cu for showing us the pliable nature and useful properties of copper.
For more information about copper see: http://www.copper.org/